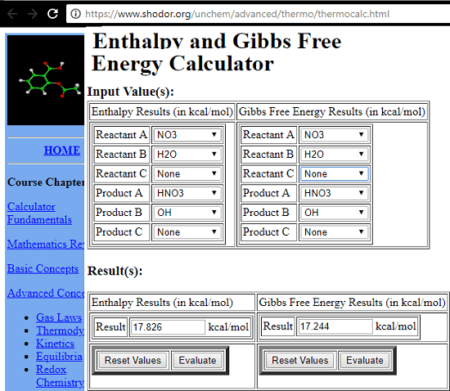
Calculate the enthalpy of formation of anhydrous Al2Cl6 from the following data: 1. 2Al(s)+6HCl(aq)——Al2Cl6(aq)+3H2(g)=1004.2kj.kj /mol 2.H2(g) +Cl2(g)—— 2Hcl(g) =184.1kj /mol 3.HCl(g)+aq—— HCl(aq)=73.2 kj/mol 4.Al2Cl6(s) + aq——– Al2Cl6(aq) =643.1 kj /mol

Calculate the standard enegry change for the reaction: OF(2)(g) +H(2)O(g) rarr O(2)(g) +2HF(g) at 298K The standard enthalpies of formation of OF(2)(g), H(2)O(g), and HF(g) are +20, -250, and -270 kJ mol^(-1),
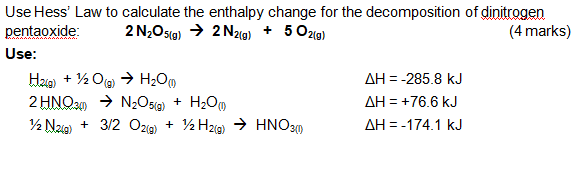
Use Hess' Law to calculate the enthalpy change for the decomposition of dinitrogen pentaoxide? | Socratic


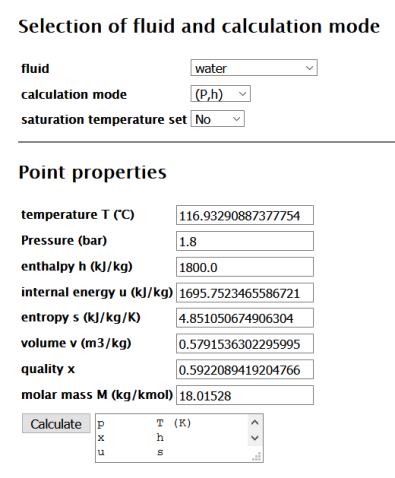


![Enthalpy Calculator - [100% Free] - Calculators.io Enthalpy Calculator - [100% Free] - Calculators.io](https://calculators.io/wp-content/uploads/2018/03/Enthalpy-Calculator.png)